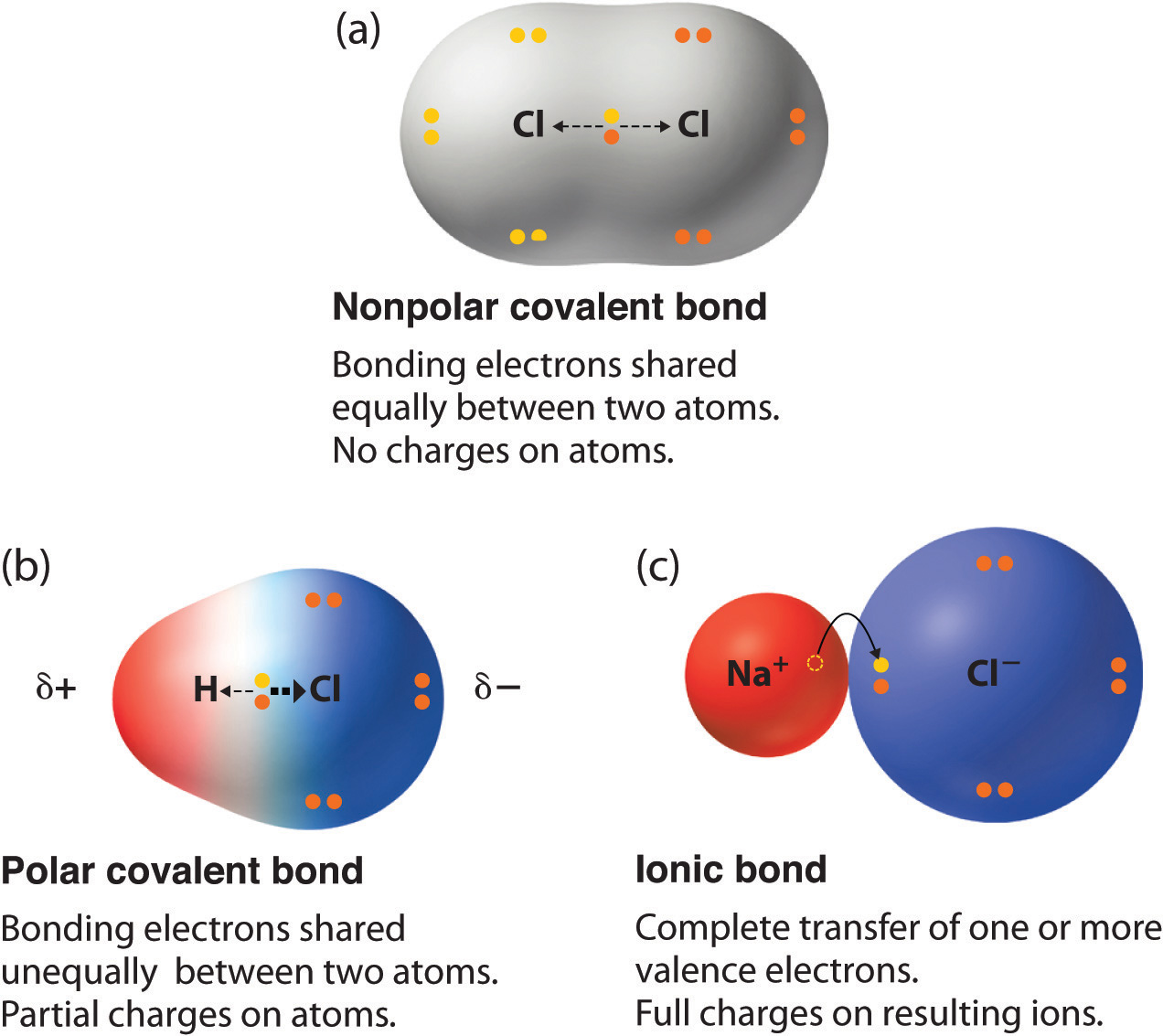
Understanding the rudimentary concepts of chemistry is essential for grasp the behavior of molecules and compounds. One such concept is the nonpolar covalent bond definition. This type of bond is spring when atoms partake electrons evenly, resulting in a poise dispersion of charge. This post will delve into the intricacies of nonpolar covalent bonds, their establishment, properties, and examples, providing a comprehensive realise of this essential chemic concept.
What is a Nonpolar Covalent Bond?
A nonpolar covalent bond is a type of chemic bond where two atoms share electrons evenly. This adequate partake results in a bond with no fond charges, making it nonpolar. The atoms affect in such bonds typically have similar negativity values, which means they have an adequate tendency to attract electrons. This balance ensures that the electrons are shared evenly, leading to a stable and inert bond.
Formation of Nonpolar Covalent Bonds
The shaping of nonpolar covalent bonds involves respective key steps:
- Electronegativity Comparison: The first step is to compare the negativity of the atoms regard. If the difference in electronegativity is minimum (usually less than 0. 5 on the Pauling scale), the bond is likely to be nonpolar.
- Electron Sharing: The atoms partake their valency electrons to achieve a stable electron configuration, typically an octet for most elements.
- Equal Distribution: The shared electrons are deal evenly between the atoms, resulting in no partial charges.
Properties of Nonpolar Covalent Bonds
Nonpolar covalent bonds exhibit several distinct properties:
- No Partial Charges: Since the electrons are shared as, there are no partial positive or negative charges on the atoms.
- Low Polarity: The lack of partial charges means that nonpolar covalent bonds have low sign, making them less responsive in polar solvents.
- Stability: These bonds are generally stable due to the poise distribution of electrons.
- Low Boiling and Melting Points: Compounds with nonpolar covalent bonds typically have lower boil and melting points compared to polar compounds.
Examples of Nonpolar Covalent Bonds
Several mutual compounds exhibit nonpolar covalent bonds. Some notable examples include:
- Hydrogen (H₂): In a hydrogen molecule, two hydrogen atoms partake their electrons as, forming a nonpolar covalent bond.
- Oxygen (O₂): Similarly, two oxygen atoms partake electrons evenly to form a nonpolar covalent bond.
- Nitrogen (N₂): Nitrogen molecules also form nonpolar covalent bonds through equal electron sharing.
- Chlorine (Cl₂): Chlorine molecules have nonpolar covalent bonds due to the adequate sharing of electrons between the chlorine atoms.
Comparing Nonpolar and Polar Covalent Bonds
To bettor realise nonpolar covalent bonds, it s helpful to compare them with polar covalent bonds. Here s a comparison:
| Property | Nonpolar Covalent Bond | Polar Covalent Bond |
|---|---|---|
| Electronegativity Difference | Less than 0. 5 | Between 0. 5 and 2. 0 |
| Electron Distribution | Equal | Unequal |
| Partial Charges | None | Present |
| Polarity | Low | High |
| Reactivity in Polar Solvents | Low | High |
Note: The negativity deviation is a key factor in regulate the type of covalent bond. A small difference indicates a nonpolar bond, while a larger difference suggests a polar bond.
Applications of Nonpolar Covalent Bonds
Nonpolar covalent bonds play a crucial role in several applications, include:
- Organic Chemistry: Many organic compounds, such as hydrocarbons, carry nonpolar covalent bonds. These compounds are essential in industries like pharmaceuticals, plastics, and fuels.
- Biochemistry: Nonpolar covalent bonds are found in biological molecules like lipids and steroids, which are crucial for cell membranes and hormone regulation.
- Materials Science: Nonpolar covalent bonds are used in the development of materials with specific properties, such as low reactivity and stability.
Challenges and Considerations
While nonpolar covalent bonds are loosely stable, there are some challenges and considerations to keep in mind:
- Reactivity: Although nonpolar covalent bonds are less responsive in polar solvents, they can still undergo reactions under certain conditions, such as eminent temperatures or in the front of catalysts.
- Environmental Impact: Some compounds with nonpolar covalent bonds, like hydrocarbons, can have important environmental impacts if not cope properly.
- Safety: Handling compounds with nonpolar covalent bonds requires careful consideration of their properties to insure safety, specially in industrial settings.
Note: Understanding the properties and behaviors of nonpolar covalent bonds is all-important for their safe and effectual use in various applications.
Nonpolar covalent bonds are a fundamental concept in chemistry, all-important for understanding the doings of molecules and compounds. By grasping the nonpolar covalent bond definition, formation, properties, and examples, one can gain a deeper discernment for the role these bonds play in various fields. From organic chemistry to biochemistry and materials science, nonpolar covalent bonds are integral to the development of new materials and technologies. By considering the challenges and applications of these bonds, we can better harness their potential while ensuring safety and environmental province.
Related Terms:
- nonpolar covalent bond definition biology
- nonpolar covalent bond examples
- polar covalent bond
- ionic bond definition
- triple covalent bond definition
- single covalent bond definition